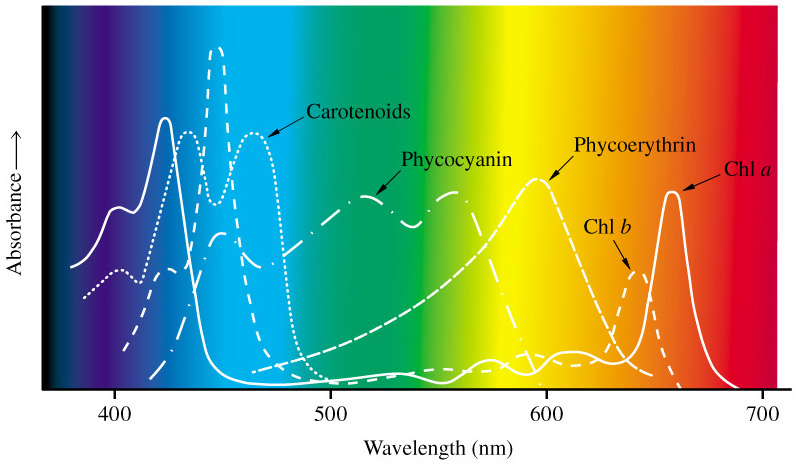
Enjoy this picture of a cheeky fellow visiting our neighbor’s tree. He is a Hooded Oriole who, with his mate, like to set up a nest in the Spring. This orange flowered tree is a favorite of the Orioles and the Hummingbirds.
Sometimes we take birds for granted. We think they’re always there, all the time. Unfortunately, songbirds are in decline due to serious predation by house cats, air pollution and various avian diseases like the West Nile virus. Habitat loss is another serious problem.
However, many bird species are seasonally occurring. Whether they are migrating through, stopping over or searching for nesting habitats you can study who’s here this month and who is coming soon. Sometimes fellow birders will tip you off that a rarity or unusual visitor has been spotted.
Local birders said there was an unusual Mexican Red Egret at the slough. It taught some of our local egrets to flap their wings while wading, herding little fishes into the shallow waters to become an easy dinner.
If you’d like to investigate your avian (bird) neighbors you’ll be more successful in the very early morning and late afternoon as birds are feeding. You may find them going about their business any time especially when their preferred food and fresh water is available. But, be careful not to set up a feeder or water dish where cats can attack. There should be a good 6’ clearance from shrubs or other cover where cats can hide.
A pair of binoculars (binocs) are very helpful and so are local bird guides.
Ask family and family friends if they are birders and can help you get started. Birders are great friends to have!
Here are some of my favorite guides and some of my favorite college level books.

Front and center: Dr. Joel Carl Welty’s famous “Life of Birds”, 3rd ed.
Left front: “Life of Birds”, 4th ed.; Dr. Welty and Dr. Baptista
Left upper row: National Geographic “Birds of North America”
Upper row, center: “Audubon Society, Field Guide to North American Birds, Western Edition”
Upper row, right: The Shorebird Guide”, O’Brien, Crossley and Karlson
Lower row, right: “The Birds of Lake Los Carneros”, Millikan and Lewis
Why am I recommending “old” editions of Dr. Welty’s book? Because it’s a classic college textbook and the old editions are more affordable! Dr. Welty was my mother’s college professor. He and his wife, Susan, were wonderful and caring people.
If you read older editions, please understand that names and classifications may change or be updated as new information is discovered, but, the beautiful line drawings are priceless. Many functions, parts or situations can’t be photographed clearly enough for viewers to understand. (I used to be a Natural Science Illustrator!)

For the new birder, the color index and color photographs in the Audubon Society Field Guides are wonderfully helpful. Buy the guide new or used for your region! Often, you’ll see photos of both male and female breeding coloration which can be somewhat different than their round the year plumage. There’s plenty of information about the range where certain birds can be found, their preferred habitat, feeding habits and lots more.
I recommend that you start an informal field journal. A simple “Composition Book” with wide-ruled lines is fine. Practice your handwriting to be legible and neat so you can write your observations with an indelible (permanent ink) pen. Keep that pen and a good number 2B drawing pencil with your Field Journal and binoculars at all times. Wildlife biologists in earlier days would have used Acid-free paper and India ink in their field journals, so you’re off to a good start!
Find a good location where you can sit, comfortably, and observe the birds in the environment around your home. Call this space your “field station” or “blind” (staying hidden so birds or animals are not disturbed by your presence).
Tell your family that you are doing Science at a particular time of the day and to please not distract you as you are taking observations.
Among the many things you may want to record is the date, time of day, weather, wind and temperature. You should try to be observing at the same time of day. Note which birds are present, note what they are doing.
Start drawing as much as you can. A quick impression is just fine, birds are rarely still.
Whether living in an apartment, in the city, in the suburbs or a more rural area, your home is essentially your habitat. You are part of the local environment. Lucky city dwellers in high-rise buildings may get to see Peregrine falcons “stooping” on pigeons, catching them for dinner. Lucky hikers can sometimes see Peregrines in the windy habitat of the Chimney Rock Trail on Point Reyes, or, see Ospreys nesting on new nest platforms along Tomales Bay, CA.
Don’t be too upset if you see a bird of prey (raptor) eating its dinner. As I say to my kids, “Everybody needs to eat.”
During my ornithology (study of birds) course at University of California Santa Cruz, I didn’t see a lot of species (types) of birds at my field site as it was late in the year (no nesting going on). I was patient. One day I saw a huge dark raptor at the top of a redwood. It could only be a Golden Eagle! My classmates doubted my observation, but on later days, I saw two Golden Eagles- one flying and another on a telephone pole at sundown.
In that class, we took field trips to the Point Reyes Bird Observatory (PRBO)and the California Academy of Sciences. This was a chance to see mist-net capture of live birds, measurement and banding of their legs at PRBO. The Academy had fixed (stuffed) bird specimens. Your local Natural Science Museum will give you the chance to see fixed specimens on display. Ask if you may sit, study and draw them!
Here are some drawings that I made of a California Condor specimen at the Carrizo Plain Visitor Center, and, sketches I did of live juvenile condors at the Santa Barbara Zoo. I guess I was hooked on Condors since my early teens when I saw a giant bird with wings wider than a picnic table flying in the wild over the Central Coast of California.

Last year, we noticed folks with binocs and cameras with long lenses staring down toward a remote beach. We stopped and these friendly birders said there are Condors on the beach! Larger than big turkeys, they were walking on the beach then flying overhead!
If you get the “birding bug” you’ll have fun the rest of your life. We have friends who travel the world trying to complete their “life lists” of birds seen and heard. You can help with the Christmas Bird Count, helping observe and tally up all the species in your area.
Thank you for joining us as we do Pandemic Projects, meant to keep you energized, curious and learning!